1. Establishment of a Domestic Hemp Production Program, US Department of Agriculture. 7 CFR §990 (2018). Accessed 3 Dec 2021. https://www.federalregister.gov/documents/2021/01/19/2021-00967/establishment-of-a-domestic-hemp-production-program
2. Gottlieb S. Statement from FDA Commissioner Scott Gottlieb, M.D., on signing of the Agriculture Improvement Act and the agency’s regulation of products containing cannabis and cannabis-derived compounds. 2018. https://www.fda.gov/news-events/press-announcements/statement-fda-commissioner-scott-gottlieb-md-signing-agriculture-improvement-act-and-agencyshttps://www.fda.gov/news-events/press-announcements/statement-fda-commissioner-scott-gottlieb-md-signing-agriculture-improvement-act-and-agencys
3. Nicolescu A, Babota M, Barros L, et al. Bioaccessibility and bioactive potential of different phytochemical classes from nutraceuticals and functional foods. Front Nutr. 2023;10:1184535. doi:10.3389/fnut.2023.1184535
4. Buxton ILO. Pharmacokinetics: The Dynamics of Drug Absorption, Distribution, Metabolism, and Elimination. In: Brunton LL, Hilal-Dandan R, Knollmann BC, eds. Goodman & Gilman's: The Pharmacological Basis of Therapeutics, 14e. McGraw-Hill Education; 2023.
5. Puckowski A, Mioduszewska K, Lukaszewicz P, et al. Bioaccumulation and analytics of pharmaceutical residues in the environment: A review. J Pharm Biomed Anal. Aug 5 2016;127:232-55. doi:10.1016/j.jpba.2016.02.049
6. Placido DF, Lee CC. Potential of Industrial Hemp for Phytoremediation of Heavy Metals. Plants (Basel). Feb 23 2022;11(5)doi:10.3390/plants11050595
7. Potter DJ. A review of the cultivation and processing of cannabis (Cannabis sativa L.) for production of prescription medicines in the UK. Drug Test Anal. Jan-Feb 2014;6(1-2):31-8. doi:10.1002/dta.1531
8. US Food and Drug Administration. What is a Botanical Drug? FDA.gov. Updated Nov 11, 2021, 2021. Accessed Mar 25, 2022. https://www.fda.gov/about-fda/center-drug-evaluation-and-research-cder/what-botanical-drug
9. Gidal BE, Vandrey R, Wallin C, et al. Product labeling accuracy and contamination analysis of commercially available cannabidiol product samples. Front Pharmacol. 2024;15:1335441. doi:10.3389/fphar.2024.1335441
10. Appendino G, Chianese G, Taglialatela-Scafati O. Cannabinoids: occurrence and medicinal chemistry. Curr Med Chem. 2011;18(7):1085-99. doi:10.2174/092986711794940888
11. VanDolah HJ, Bauer BA, Mauck KF. Clinicians' Guide to Cannabidiol and Hemp Oils. Mayo Clin Proc. Sep 2019;94(9):1840-1851. doi:10.1016/j.mayocp.2019.01.003
12. Mackie K. Cannabinoid receptors: where they are and what they do. J Neuroendocrinol. May 2008;20 Suppl 1:10-4. doi:10.1111/j.1365-2826.2008.01671.x
13. Hillig KW, Mahlberg PG. A chemotaxonomic analysis of cannabinoid variation in Cannabis (Cannabaceae). Am J Bot. Jun 2004;91(6):966-75. doi:10.3732/ajb.91.6.966
14. Small E. Evolution and Classification of Cannabis sativa (Marijuana, Hemp) in Relation to Human Utilization. The Botanical Review. Sept 1 2015;81(3):189-294. doi:10.1007/s12229-015-9157-3
15. Mikos RA, Kam CD. Has the "M" word been framed? Marijuana, cannabis, and public opinion. PLoS One. 2019;14(10):e0224289. doi:10.1371/journal.pone.0224289
16. Mekonnen S. Foxglove -Toxic to the Heart. National Capital Poison Center. Accessed Oct 21, 2022. https://www.poison.org/articles/foxglove
17. US Food and Drug Administration. Remarks by Lowell Schiller, JD at the Council for Responsible Nutrition Conference. 2019. Accessed Oct 21, 2022. https://www.fda.gov/news-events/speeches-fda-officials/remarks-lowell-schiller-jd-council-responsible-nutrition-conference-1172019-11072019
18. Potter D. The Propagation Characterisation and Optimisation of Cannabis Sativa L as a Phytopharmaceutical. King's College London; 2009.
19. Morales P, Hurst DP, Reggio PH. Molecular Targets of the Phytocannabinoids: A Complex Picture. Prog Chem Org Nat Prod. 2017;103:103-131. doi:10.1007/978-3-319-45541-9_4
20. Udoh M, Santiago M, Devenish S, McGregor IS, Connor M. Cannabichromene is a cannabinoid CB(2) receptor agonist. Br J Pharmacol. Dec 2019;176(23):4537-4547. doi:10.1111/bph.14815
21. Abioye A, Ayodele O, Marinkovic A, Patidar R, Akinwekomi A, Sanyaolu A. Delta-9-tetrahydrocannabivarin (THCV): a commentary on potential therapeutic benefit for the management of obesity and diabetes. J Cannabis Res. Jan 31 2020;2(1):6. doi:10.1186/s42238-020-0016-7
22. Rosenberg EC, Tsien RW, Whalley BJ, Devinsky O. Cannabinoids and Epilepsy. Neurotherapeutics. Oct 2015;12(4):747-68. doi:10.1007/s13311-015-0375-5
23. Hill AJ, Mercier MS, Hill TD, et al. Cannabidivarin is anticonvulsant in mouse and rat. Br J Pharmacol. Dec 2012;167(8):1629-42. doi:10.1111/j.1476-5381.2012.02207.x
24. US Food and Drug Administration. FDA Approves First Drug Comprised of an Active Ingredient Derived from Marijuana to Treat Rare, Severe Forms of Epilepsy. 2018. Accessed Oct 21, 2022. https://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-comprised-active-ingredient-derived-marijuana-treat-rare-severe-forms
25. Tahir MN, Shahbazi F, Rondeau-Gagne S, Trant JF. The biosynthesis of the cannabinoids. J Cannabis Res. Mar 15 2021;3(1):7. doi:10.1186/s42238-021-00062-4
26. Bolognini D, Rock EM, Cluny NL, et al. Cannabidiolic acid prevents vomiting in Suncus murinus and nausea-induced behaviour in rats by enhancing 5-HT1A receptor activation. Br J Pharmacol. Mar 2013;168(6):1456-70. doi:10.1111/bph.12043
27. Umpierrez LS, Costa PA, Michelutti EA, et al. Cannabidiol but not cannabidiolic acid reduces behavioural sensitisation to methamphetamine in rats, at pharmacologically effective doses. Psychopharmacology (Berl). May 2022;239(5):1593-1603. doi:10.1007/s00213-022-06119-3
28. Pagano C, Navarra G, Coppola L, Avilia G, Bifulco M, Laezza C. Cannabinoids: Therapeutic Use in Clinical Practice. Int J Mol Sci. Mar 19 2022;23(6):3344. doi:10.3390/ijms23063344
29. Anderson LL, Heblinski M, Absalom NL, et al. Cannabigerolic acid, a major biosynthetic precursor molecule in cannabis, exhibits divergent effects on seizures in mouse models of epilepsy. Br J Pharmacol. Dec 2021;178(24):4826-4841. doi:10.1111/bph.15661
30. Janecki M, Graczyk M, Lewandowska AA, Pawlak L. Anti-Inflammatory and Antiviral Effects of Cannabinoids in Inhibiting and Preventing SARS-CoV-2 Infection. Int J Mol Sci. Apr 10 2022;23(8):4170. doi:10.3390/ijms23084170
31. Weydt P, Hong S, Witting A, Moller T, Stella N, Kliot M. Cannabinol delays symptom onset in SOD1 (G93A) transgenic mice without affecting survival. Amyotroph Lateral Scler Other Motor Neuron Disord. Sep 2005;6(3):182-4. doi:10.1080/14660820510030149
32. Federal Food, Drug, and Cosmetic Act: Subchapter II - Definitions, US Food and Drug Administration. 21 USC §321 (2024). https://uscode.house.gov/view.xhtml?req=granuleid:USC-prelim-title21-section321&num=0&edition=prelim
33. US Food and Drug Administration. FDA Regulation of Cannabis and Cannabis-Derived Products, Including Cannabidiol (CBD). Updated Jul 5, 2023. Accessed Jul 13, 2023. https://www.fda.gov/news-events/public-health-focus/fda-regulation-cannabis-and-cannabis-derived-products-including-cannabidiol-cbd
34. US Drug Enforcement Administration. Drug Scheduling. Updated Jul 10, 2018. Accessed Dec 4, 2021. https://www.dea.gov/drug-information/drug-scheduling
35. Castillo PE, Younts TJ, Chavez AE, Hashimotodani Y. Endocannabinoid signaling and synaptic function. Neuron. Oct 4 2012;76(1):70-81. doi:10.1016/j.neuron.2012.09.020
36. Appendino G, Chianese G, Taglialatela-Scafati O. Cannabinoids: Occurence and Medicinal Chemistry. Current Medicinal Chemistry. 2011;18:1085-1099. doi:10.2174/092986711794940888
37. Sugiura T, Kondo S, Sukagawa A, et al. 2-Arachidonoylglycerol: a possible endogenous cannabinoid receptor ligand in brain. Biochem Biophys Res Commun. Oct 4 1995;215(1):89-97. doi:10.1006/bbrc.1995.2437
38. Mechoulam R, Ben-Shabat S, Hanus L, et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem Pharmacol. Jun 29 1995;50(1):83-90. doi:10.1016/0006-2952(95)00109-d
39. Ben-Shabat S, Fride E, Sheskin T, et al. An entourage effect: inactive endogenous fatty acid glycerol esters enhance 2-arachidonoyl-glycerol cannabinoid activity. Eur J Pharmacol. Jul 17 1998;353(1):23-31. doi:10.1016/s0014-2999(98)00392-6
40. US Food and Drug Administration. Development & Approval Process Drugs. 2019. Accessed Dec 5, 2021. https://www.fda.gov/drugs/development-approval-process-drugs
41. Legitimacy of Industrial Hemp Research, Government US. 7 CFR §7606 (2014).
42. Small E, Marcus D. Hemp: A New Crop with New Uses for North America. In: Janick J, Whipkey A, American Society for Horticultural Science, eds. Trends in New Crops and New Uses. ASHS Press; 2002:284-326.
43. Department of Justice, Drug Enforcement Administration. (2020) Marijuana/Cannabis Fact Sheet. https://www.dea.gov/sites/default/files/2020-06/Marijuana-Cannabis-2020_0.pdf
44. US Food and Drug Administration. FDA and Cannabis: Research and Drug Approval Process. Updated Oct 10, 2020. Accessed Sep 2, 2021. https://www.fda.gov/news-events/public-health-focus/fda-and-cannabis-research-and-drug-approval-process
45. Cash MC, Cunnane K, Fan C, Romero-Sandoval EA. Mapping cannabis potency in medical and recreational programs in the United States. PLoS One. 2020;15(3):e0230167. doi:10.1371/journal.pone.0230167
46. Bostwick JM. Blurred boundaries: the therapeutics and politics of medical marijuana. Mayo Clin Proc. Feb 2012;87(2):172-86. doi:10.1016/j.mayocp.2011.10.003
47. Marijuana Policy Project. “Prescribing” Versus “Recommending" Medical Cannabis. 2021. Accessed Dec 3, 2022. https://www.mpp.org/issues/medical-marijuana/prescribing-versus-recommending-medical-cannabis/
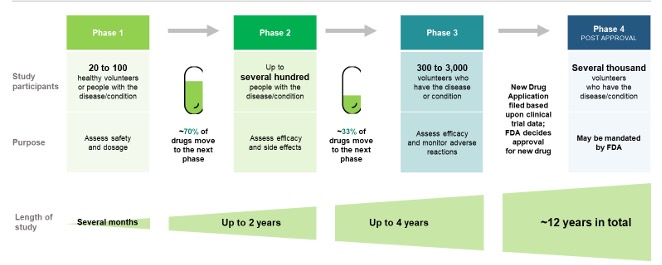
48. US Food and Drug Administration. Basics About Clinical Trials. FDA.gov. 2023. Accessed Oct 1, 2024. https://www.fda.gov/patients/clinical-trials-what-patients-need-know/basics-about-clinical-trials
49. Koch A, Brandenburger S, Türpe S, Birringer M. The Need for a Legal Distinction of Nutraceuticals. Food and Nutrition Sciences. 2014;05(10):905-913. doi:10.4236/fns.2014.510100
50. Ojuederie OB, Babalola OO. Microbial and Plant-Assisted Bioremediation of Heavy Metal Polluted Environments: A Review. Int J Environ Res Public Health. Dec 4 2017;14(12)doi:10.3390/ijerph14121504
51. Wieten S. Expertise in evidence-based medicine: a tale of three models. Philos Ethics Humanit Med. Feb 2 2018;13(1):2. doi:10.1186/s13010-018-0055-2
52. Devereaux PJ, Yusuf S. The evolution of the randomized controlled trial and its role in evidence-based decision making. J Intern Med. Aug 2003;254(2):105-13. doi:10.1046/j.1365-2796.2003.01201.x
53. US Food and Drug Administration. (2022) Submitting Documents using RWD and RWE to FDA for Drug and Biological Products. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/submitting-documents-using-real-world-data-and-real-world-evidence-fda-drug-and-biological-products
54. Curran HV, Morgan CJA. Desired and Undesired Effects of Cannabis on the Human Mind and Psychological Well-Being. In: Pertwee R, ed. Handbook of Cannabis. Oxford University Press; 2014:chap 36.
55. Roque-Bravo R, Silva RS, Malheiro RF, et al. Synthetic Cannabinoids: A Pharmacological and Toxicological Overview. Annu Rev Pharmacol Toxicol. Jan 20 2023;63:187-209. doi:10.1146/annurev-pharmtox-031122-113758
56. National Institute on Drug Abuse. Synthetic Cannabinoids. National Institutes of Health. Updated Oct 2023, 2023. Accessed Oct 1, 2024. https://nida.nih.gov/publications/drugfacts/synthetic-cannabinoids-k2spice
57. Tanney CAS, Backer R, Geitmann A, Smith DL. Cannabis Glandular Trichomes: A Cellular Metabolite Factory. Front Plant Sci. 2021;12:721986. doi:10.3389/fpls.2021.721986
58. Tagen M, Klumpers LE. Review of delta-8-tetrahydrocannabinol (Delta(8) -THC): Comparative pharmacology with Delta(9) -THC. Br J Pharmacol. Aug 2022;179(15):3915-3933. doi:10.1111/bph.15865
59. Turner CE, Elsohly MA, Boeren EG. Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J Nat Prod. Mar-Apr 1980;43(2):169-234. doi:10.1021/np50008a001
60. Sirikantaramas S, Taura F, Tanaka Y, Ishikawa Y, Morimoto S, Shoyama Y. Tetrahydrocannabinolic acid synthase, the enzyme controlling marijuana psychoactivity, is secreted into the storage cavity of the glandular trichomes. Plant Cell Physiol. Sep 2005;46(9):1578-82. doi:10.1093/pcp/pci166
61. Dayanandan P, Kaufman PB. Trichomes of Cannabis Sativa L. (Cannabaceae). American Journal of Botany. 1976;63(5):578-591. doi:10.1002/j.1537-2197.1976.tb11846.x
Sponsored by Jazz Pharmaceuticals, Inc.
For further information, please access https://www.jazzpharma.com.
©2024, Jazz Pharmaceuticals, Inc.